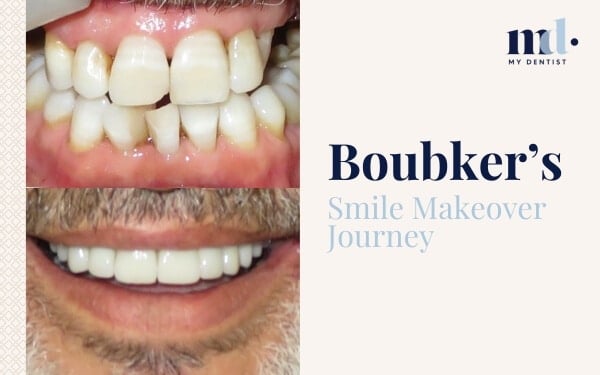
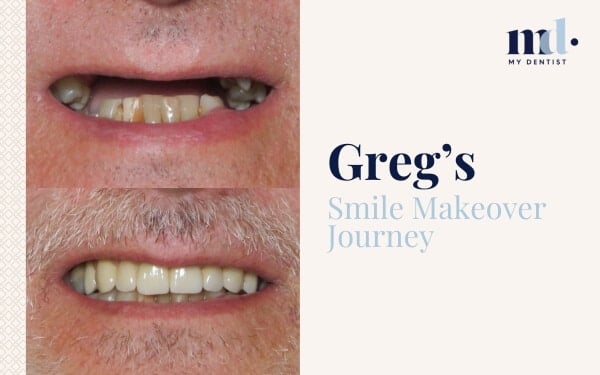
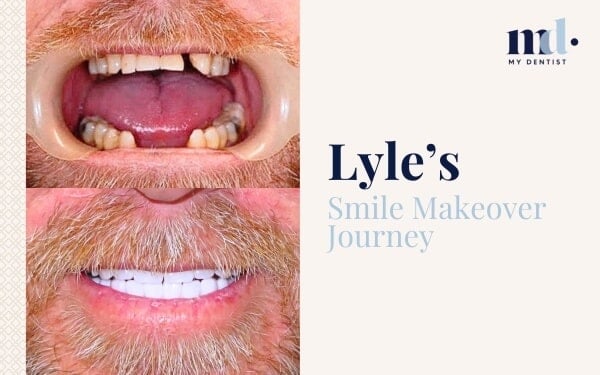
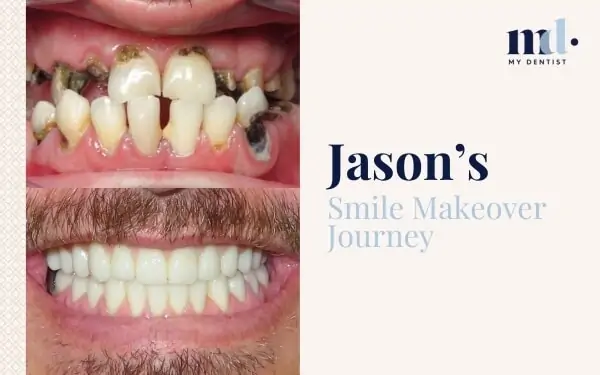
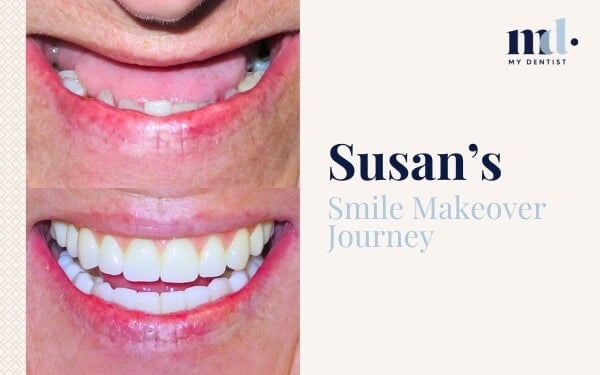
Now There’s A Score For Gum Inflammation For Better Treatment Outcomes Alderley, Newmarket, Brisbane
We’re living amid a golden age of scientific discovery.
Often we’re told that not all that glitters is gold; and we usually anticipate that the goose that lays the golden egg will entice a wild chase first.
It’s not so different for scientists.
Lead in part by recent technological advances in next-generation sequencing (NGS) the wearying weight of all collected, and accruing, data brings its own challenges.
In 2022, researchers looked closely at periodontitis, a chronic inflammatory disease caused by the interaction of oral microorganisms with the host immune response.
And it’s complicated.
Derived from the Greek ‘cyto’ meaning ‘cell’ and ‘kinos’ – ‘movement’ – cytokines often act as first responders to pathogens contaminating the immune system.
Porphyromonas gingivalis (P.G.) for example, is a key mediator in disrupting the self-regulating process of maintaining cell stability while adjusting to changing conditions.
P.g. impedes phagocytosis: a biological process for ingesting and eliminating particles of a particular size, where a cell binds to the item it wants to destroy, and engulfs it. Cells can be foreign substances, micro-organisms or apoptotic – those hitting the program for cell death.
At the same time it’s inhibiting a defense mechanism, P.g. kills the protective capacity of immune cells by releasing and increasing selected cytokines.
Often described as a ‘cytokine storm’ (or cytokine release syndrome) the immune system responds to infection more aggressively than it should, producing too many anti-inflammatory messages. Cells discharge these proteins to signal to other cells that a viral invasion is in progress. The diversity of these cytokines is expected to exceed one hundred – and each one calls for a specific biological response.
We’re pretty much all special little snowflakes when it comes to our immune system. With the unraveling and understanding of cytokines, a crucial issue is that to date, there is no definition of what constitutes a ‘normal’ range.
Everyone’s is different. How disease affects an individual can vary greatly.
That goes for medications as well.
Globally, although pharmaceutical companies hold data on millions of individuals, what makes determining a representation of typical cytokine levels impracticable is because every variable counts.
Sex, age, ethnicity and the sample time of day are a snapshot of the kinds of impacts that can skew results.
The reason science is so interested in pinning all this down, is that when cytokines thresholds are breached, it’s a marker for disease. So much so, it’s predicted to be eventually included in all standard blood tests.
A cytokine (of which there are many) is a signal that never occurs alone – only, and always, it is part of a complex milieu.
It connects and interacts with lipid moieties: that which has two parts and can be divided.
Nucleotides, growth factors, neuropeptides, alarmins, hormones, and microbial reagents are part of the cytokine circle of influence. The focus of research is how cytokines engage and interlock with the many systems involved in maintaining cell integrity and therefore good health.
Fortunately, cytokines are also found in saliva, making it easier to sample. One study wanted to know if clinically detected gum inflammation could predict the level of cytokines found in saliva.
Salivary cytokines look into the molecular composition of the oral environment.
What was demonstrated is that a single score – that encompasses several salivary cytokines – is in parallel with the severity of the periodontal inflammation.
Cytokinology is an investigational. Hypotheses are now being questioned and challenged. New tests are constantly being devised because of its complex triggers and resulting interlaced network of chronic, negative impact.
Researchers caution that more study is needed to validate the cytokine score in patients with different health conditions, and levels of periodontal disease – including early-stage, and healthy.
If the cytokine score is substantiated in larger, more diverse populations, it could be used to better understand periodontal disease progression and recurrence as well as more specific connections to other systemic conditions.
Recently, researchers have raised timing as an issue in suppressed immune systems. Too soon after infection could possibly thwart initial antiviral responses; too late risks immunity overreaction.
It’s information useful for drug administration when immune responses need to be curbed.
Certainly there’s strong confirmation that gum disease is associated with a widening range of health conditions.
Before research confirmed links with oral bacteria, and conditions like osteoporosis, rheumatoid arthritis, kidney and liver disease, as well as diabetes and heart disease – there were decades of not knowing whether regular dental care and periodontal treatments prevented or even improved other diseases.
There is the possibility too, that the cytokine network is significantly affected by oral microbiome.
From that perspective, it could very well be a cybernetic loop between them, where each is an infinite feedback of the other. That it therefore may be possible to treat, or even eradicate, gum disease by altering oral microbiota is an incredibly enlightening thought.
What science knows at this point is that persistent microbial stimulation optimises immune response that results in the induction of cytokines.
Along with that is the considerable evidence that viruses produce many cytokine mimics to gain advantage. After all, they are a major driver in human development.
Whether or not bacteria do the same has not been as extensively documented, but it’s a reasonable expectation that evolution will have created microorganisms that directly respond to cytokines, or genes that originate from within an organism, encoding cytokine mimics and infiltrators.
The inside of your mouth is the perfect place for bacteria to thrive. It’s dark, it’s wet, it’s warm. Everything that goes into it (everything!) provides nutrients (or not) for the you that you see – and the massively bigger, symbiotic, microbiota invisible-you that you don’t.
When harmful bacteria build up around teeth and gums, you’re at risk of developing conditions in your mouth that influence the rest of your body.
Researchers have some evidence to suggest that periodontal bacteria from the mouth can travel to the arteries in vascular disease patients, potentially playing a role in the development of the disease.
Basically, compromised health is all about inflammation – it’s the clever flashing red light of the immune system letting the body know that something’s amiss.
Smoking is a packed factor in the immune response of periodontitis patients. It’s clearly accepted that (among other things) it changes the microflora; what’s yet to be determined is in what exact capacity.
Non-smoking and smoking periodontitis patients had their cytokine profiles taken both before, and after periodontal therapy, in order to ascertain its influence on the disease.
The baseline cytokine profiles showed no significant difference between the smoking and non-smoking patients.
However after treatment, smokers showed significantly higher levels of the cytokine IL-1β than non-smokers – indicating that smoking impairs the work being done by periodontal therapies.
To clinically measure the level of periodontal inflammation, researchers used a formula.
Called the Periodontal Inflamed Surface Area (PISA), it’s calculated using depth-of-pocket measurements of the gums, and the level of probed bleeding. PISA is a single measure of periodontal inflammation: the higher the score, the worse the inflammation.
What was found is that PISA scores are significantly associated with new cytokine scores – independent of factors including age, gender, smoking, and body mass index (BMI). The higher a cytokine score, the more severe the periodontal inflammation.
It’s acknowledged that the PISA score reduces with scaling and planing treatment.
What’s interesting is whether the cytokine score also drops; and if it doesn’t, why?
Is it registering an underlying systemic inflammation? Does the hyper-inflammatory response of a high cytokine score predict the recurrence of periodontitis?
These are the questions driving this ongoing research.
Over the last decade, the world of host and pathogen genetics has been revolutionised. The cytokine storm has captured the attention of the scientific community. While the concept of excessive, or uncontrolled release of pro-inflammatory cytokines is understood in terms of infectious and non-infectious disease, the biological consequences are yet to be clearly defined.
Through increasingly sophisticated biostatistical analysis, exactly what associations mean, and how they should be interpreted will continue to be developed.
And that, is golden.
The content has been made available for informational and educational purposes only. My Dentist does not make any representation or warranties with respect to the accuracy, applicability, fitness, or completeness of the content.
The content is not intended to be a substitute for professional personal diagnosis or treatment. Always seek the advice of your dentist or another qualified health provider with any questions you may have regarding a dental or medical condition. Never disregard professional advice or delay seeking it because of something you have read or seen on the Site.
Services We Mentioned:
General Dentistry
Essential care for healthy smiles — checkups, cleanings, and treatments for overall oral wellness and lasting health.
Periodontal Treatment for Gum Disease
Restoring gum health, preventing tooth loss, and preserving a strong, healthy smile through specialised care.